International Journal of Pharmaceutical Sciences and Developmental Research
Repaglinide-Indinavir Pharmacokinetic and Pharmacodynamic Interaction: Non-Clinical Evaluation in Hepatic and Diabetic Impairment models
1Acubiosys Private Limited, Telangana State Industrial Infrastructure Corporation Limited - Industrial Area Local Authority, Moula Ali, Hyderabad, Telangana 500040, India
2Chebrolu Hanumaiah institute of pharmaceutical sciences, Chowdavaram, Guntur, Andhra pradesh 522019, India
Author and article information
Cite this as
Jeevan Karthik M, Aakarsha T, Lokesh J, Manaswini C, Rameshbabu J, Srinivas M. Repaglinide-Indinavir Pharmacokinetic and Pharmacodynamic Interaction: Non-Clinical Evaluation in Hepatic and Diabetic Impairment models. Int J Pharm Sci Dev Res. 2026; 12(1): 9-16. Available from: 10.17352/ijpsdr.000059
Copyright License
© 2026 Srinivas M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Background: Repaglinide, used for postprandial glucose control, is extensively metabolized by cytochrome P450 enzymes and transported by OATP and P-glycoprotein, making it susceptible to pharmacokinetic interactions. Indinavir, an antiviral protease inhibitor, affects metabolic and transport pathways, thereby altering the pharmacokinetic and pharmacodynamic profile of repaglinide. This study investigated the interaction between repaglinide and indinavir in healthy rats and in rat models of diabetes and hepatic impairment.
Methods: Clinical doses of indinavir and repaglinide were converted to rat-equivalent doses using the Body Surface Area (BSA) method. Healthy, diabetic, and hepatic-impaired rats were administered indinavir (78 mg/kg, P.O), followed by repaglinide (0.5 mg/kg, P.O). Plasma concentrations of repaglinide were determined, and pharmacokinetic parameters, including maximum plasma concentration (Cmax), Area Under the Concentration–time curve (AUC), elimination half-life (t½), and plasma clearance (CL), were evaluated. Blood glucose levels were also measured to assess pharmacodynamic effects.
Results: In the presence of indinavir, repaglinide exhibited significantly altered pharmacokinetics in normal (P < 0.005), diabetic (P < 0.0001), and hepatic-impaired (P < 0.0001) rats, characterized by increased maximum plasma concentration (Cmax), Area Under the Concentration–time curve (AUC), and elimination half-life (t½), along with a significant reduction in plasma clearance (CL) across all groups compared with repaglinide alone. Pharmacodynamically, repaglinide in combination with indinavir produced a markedly greater (P < 0.0001) hypoglycemic effect in diabetic and hepatic-impaired rats than repaglinide administered alone.
Conclusion: The study demonstrates a significant pharmacokinetic and pharmacodynamic interaction between indinavir and repaglinide, characterized by enhanced bioavailability and reduced total body clearance of repaglinide. These effects are likely mediated by the inhibitory action of indinavir on cytochrome P450 enzymes, as well as on OATP and P-glycoprotein transporters.
Drug metabolism generally involves a number of different enzyme systems and pathways. Most drug metabolic reactions are catalyzed by one or multiple cytochrome P-450 isoforms. Prescribing drugs for hepatic impairment is a critical and complex component of clinical practice. Liver disease exerts variable influences on pharmacokinetic and pharmacodynamic processes, notably altering plasma protein binding, which subsequently impacts the distribution and elimination of numerous drugs [1]. HIV patients treated with combinations of antiretroviral therapy (HAART) regimens experience a significant reduction in plasma HIV viral load, and restoration of CD4+ lymphocyte counts contributes to the deceleration of disease progression and enhancement of patient survival outcomes [2]. Protease Inhibitors (PIs) based therapy has metabolic complications, including Glucose intolerance, diabetes mellitus, and insulin resistance. Long-term treatment of HIV can lead to hepatic dysfunction in HIV patients. A major challenge faced by healthcare clinicians treating patients with hepatic impairment, HIV infection is a complicated problem due to medication interactions that are associated with highly active antiretroviral treatment – HAART [3]. The simultaneous use of multiple therapeutic agents increases the potential for drug interactions. Such drug-drug interactions are especially concerning in patients with AIDS, as these individuals frequently necessitate multiple pharmacological agents, including antiretroviral therapies and medications targeting opportunistic infections and related comorbidities. Consequently, the risk of drug interactions constitutes a critical factor to consider in the management of treatment involving HIV protease inhibitors [4]. Repaglinide is a meglitinide class of new, rapid-acting prandial oral hypoglycaemic agents improved and developed for the therapy of patients with type 2 diabetes. Repaglinide exhibits the binding affinity towards the sulfonylurea binding pocket on pancreatic β-cells & has a similar mode of action to that of sulfonylureas, but it displays specific therapeutic attributes compared to sulfonylureas. After administration, repaglinide is quickly absorbed, with peak concentrations occurring within 1 hour of an individual oral dose of 2mg of repaglinide [5]. Repaglinide exhibits high plasma protein binding (>98 %). It is quickly removed from the bloodstream, with a terminal elimination half-life (t1/2) of less than 1 hour. Repaglinide plasma concentrations decrease quickly, attaining pre-dose levels within 4 or 5 hours after oral intake of 2 mg of the drug [6]. Repaglinide is metabolized in the liver to inactive metabolites via the CYP3A4 enzyme system. It is primarily metabolized through oxidative biotransformation via the hepatic cytochrome P450 system, particularly with the CYP3A4 and CYP2C8 isoforms, and inactive metabolites are excreted mainly in the faeces. Repaglinide has an affinity for P-gp and OATP transporters and can significantly contribute to potential drug-drug interactions with other P-gp and OATP substrates or inhibitors [7]. Indinavir is primarily metabolized by the liver, and in vitro studies indicate that cytochrome P-450 3A4 (CYP3A4) is the major enzyme responsible for the formation of the oxidative metabolites. The co-administration of indinavir with other medications predominantly metabolized by CYP3A4 may result in elevated plasma concentrations of the other drugs, which could increase or prolong their therapeutic effects and may cause adverse effects. Based on in vitro data in human liver microsomes, indinavir is an inhibitor of the cytochrome P450 isoform CYP3A4 and a weak inhibitor of CYP2D6. The impact of indinavir on the 6β-hydroxylation of testosterone catalyzed by the microsomal fraction of human liver has been extensively studied. Kinetic studies revealed that indinavir acts as a reversible inhibitor of testosterone 6β-hydroxylase, with a Ki value indicating its inhibitory potential toward CYP3A4 metabolized drugs. Additionally, the involvement of P-glycoprotein in regulating the cerebral accumulation of indinavir was confirmed through an interaction study with cyclosporine in rat models. Concomitant administration of cyclosporine resulted in a more than threefold increase in indinavir concentrations in rats. A large fraction of pharmacokinetic drug interactions observed in HIV therapy is primarily caused by changes in ADME of either the HIV drug itself or the concurrently administered medication. They may involve alterations in drug metabolism mediated by the CYP-450 system, modulation of P-gp, and changes in drug elimination by hepatic or renal route [8].
The present study hypothesized that the elimination of repaglinide might be affected by CYP3A4 enzyme and transporter-mediated inhibition. The cytochrome P450 enzyme system metabolizes Repaglinide and serves as a substrate for the CYP3A4 enzyme, as well as the P-glycoprotein (P-gp) and Organic Anion-Transporting Polypeptide (OATP) transporters. Indinavir, an inhibitor of CYP3A4 enzyme, OATP, and P-gp transporters, might have altered the pharmacokinetics of repaglinide in vivo. Since the PK/PD drug-drug interaction between repaglinide and indinavir at therapeutic doses had not been reported, this study investigated the effects of indinavir on the pharmacokinetics and pharmacodynamics of repaglinide in healthy, diabetic, and liver-dysfunction rat models.
Materials and methods
Chemicals
Repaglinide was gifted as a gift sample from Dr. Reddy’s Laboratories (Hyderabad, India), while Indinavir was procured from AurobindoPharma Limited (Hyderabad, India). Alloxan monohydrate was purchased from Sigma-Aldrich (Bangalore, India). Glucose estimation kits were procured from Agappe Diagnostics (Mumbai, India). HPLC-grade acetonitrile and formic acid were obtained from Merck Chemicals (Mumbai, India).
Instrumentation
An Agilent Technologies HPLC system (California, USA) coupled with an API-3200MS/MS mass spectrometer Sciex Technologies (Foster City, CA, USA), and equipped with a Hypersil GOLD column, C18, 5µm, 50*4.6mm internaldiameter (Thermo Scientific) was used for chromatographic separation and integration of the analytes.
Methods
Pharmacokinetics and Pharmacodynamics Interaction Study: Male albino Wistar rats, with a bodyweight ranging from 200-250 g, were procured from Jeeva Life Sciences (Hyderabad, India).To understand the conditions, the animals were housed at Jeeva Life Sciences, Hyderabad, under a 12-hour light/dark cycle. The rats were fasted overnight before dosing and for 4 hours following dosing. Water was provided ad libitum throughout the deprivation period. Prior approval of the experimental design was issued by the institutional animal ethics committee (1305/ac/09/CPCSEA). For Control and Supervision of Experiments and Animals (CPCSEA), the experiments were conducted according to the guidelines provided by the committee.Ratsweredividedinto4 groups (n=6 per group): Group 1, Group 2 consisted of healthy rats, Group 3 included diabetic rats, and Group 4 comprised hepatically impaired rats. Groups 2, 3, and 4 received indinavir at a dosage of 78 mg/kg. After an interval of 30 minutes, repaglinide was administered at a dosage of 0.5 mg/kg to all four groups [9,10]. The oral dosing formulation was prepared using the gravimetric dilution method with 1% Tween-80 and 0.5% methylcellulose as the vehicle [11].
Induction of diabetes: Diabetes was experimentally established in rats through the intraperitoneal administration of alloxan monohydrate dissolved in ice-cold normal saline at doses of 100 mg/kg and 50 mg/kg body weight, given consecutively over two days [12]. At 72 hours post administration, blood samples were withdrawn from the surviving rats via retro-orbital puncture, and plasma glucose concentration was analyzed. Animals with circulating glucose concentration equal to or exceeding 200 mg/dL were selected for the study [13].
Induction of hepatic impairment: Hepatic impairment in rats was experimentally induced through the intraperitoneal administration of a carbon tetrachloride and olive oil mixture, combined in equal proportions, at a dosage of 2 mL/kg for a duration of one day [14]. After 24 hours, blood samples were collected from rats by retro-orbital puncture of all surviving animals, and the serum was analyzed for elevated total bilirubin (>2mg/dL), alanine transaminase (>150 mg/dL), aspartate transaminase (>200mg/dL), and albumin (<3 mg/dL) were selected for the study [15].
The blood samples were obtained by retro-orbital puncture at the subsequent time points: 0, 0.25, 0.5, 1, 2, 4, 6, 8, 12, and 24h from all animal groups in duplicate. For plasma collection, blood samples were drawn into EDTA-coated tubes, while serum samples were collected in regular tubes. These samples were then centrifuged at 8000rpm for 10 minutes. The supernatant was transferred into vials and stored at -80 °C. Plasma samples were analyzed using LC/MS-MS, while serum samples were used for blood glucose estimation at respective time points [12].Liquid chromatography-mass spectrometry method: The repaglinide concentrations in rat plasma samples were determined using a validated liquid Chromatography/Mass Spectrometry (LC-MS/MS) technique. Positive-ion multiple reaction monitoring enabled the tandem mass spectrometric detection of repaglinide, indinavir, and rosuvastatin. The selected precursor ions were [M+H] + at m/z 453.2 for repaglinide and m/z 482.3 for rosuvastatin, with corresponding fragment ions monitored at m/z 230.3 and 258.15 for repaglinide and rosuvastatin (internal standard), respectively. The chromatographic separation was achieved using a Hypersil GOLD C18 column (4.6 x 100 mm, 5 µm), maintained at 40 °C with a flow rate of 1.3 mL/min. The mobile phase consisted of 10 mM ammonium acetate and acetonitrile, utilizing isocratic elution in a 50:50 ratio and an overall run time of 6.0 minutes. In vivo, samples were prepared through protein precipitation by adding 200 µL of acetonitrile spiked with the internal standard, rosuvastatin, to 50 µL of the sample. The samples were vortexed and centrifuged at 4000 rpm for 10 minutes before evaporating the supernatant. The developed method was validated following the International Conference on Harmonization [16].
Pharmacokinetic profiling and statistical analysis: Pharmacokinetic parameters were obtained by fitting plasma concentration-time data to a non-compartmental model utilizing Phoenix software (v6.3.0.395; Pharsight, Mountain View, CA). The maximum plasma concentration (Cmax) and the time to reach Cmax (Tmax) were analyzed, while the elimination half-life (t½) was determined by dividing 0.693 by the slope derived from the log-linear regression of the terminal phase of the plasma concentration profile. The area under the concentration-time curve from time zero to the last measurable concentration (AUC0-t) and extrapolated to infinity (AUC0-∞) were calculated employing the linear/log trapezoidal method. Total plasma clearance (CL) was also estimated using the Phoenix software. Data are expressed as mean ± standard deviation (SD), and statistical significance was assessed via one-way Analysis Of Variance (ANOVA), followed by Dunnett’s test with a threshold of p < 0.0001 considered indicative of significance.
Estimation of glucose: The glucose concentrations were determined by using the glucose oxidase peroxidase method, following the instructions provided with the commercial kits. The assay utilized glucose (S.L) R1 reagent, which consisted of tris buffer, phenol, glucose oxidase, and 4-amino phenazone, along with a glucose standard solution (100mg/dL). Assay blank, standard, and test samples were prepared by mixing 1 ml of glucose (S.L) R1 reagent with 10µl distilled water, glucose standard, and plasma sample. Upon reaction, a colored complex was formed, and the intensity of the developed color was measured by a colorimeter at 530 nm.
Eq. (1) Relative decrease in BGL = ((IBGL– FBGL) / IBGL) x 100
BGL stands for blood glucose level, IBGL indicates the Baseline blood glucose level, and FBGL represents the Post blood glucose level [17].
Results and discussion
Bioanalytical validation of the LC-MS/MS Method
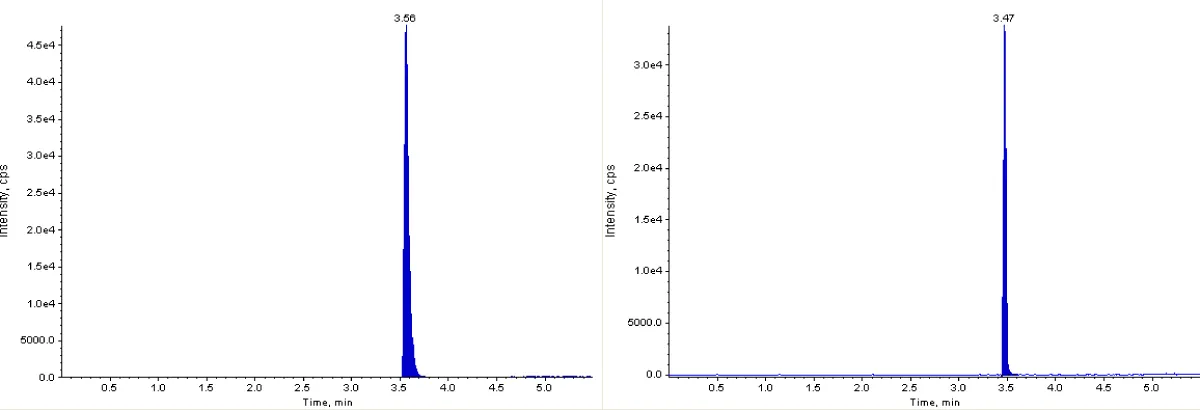
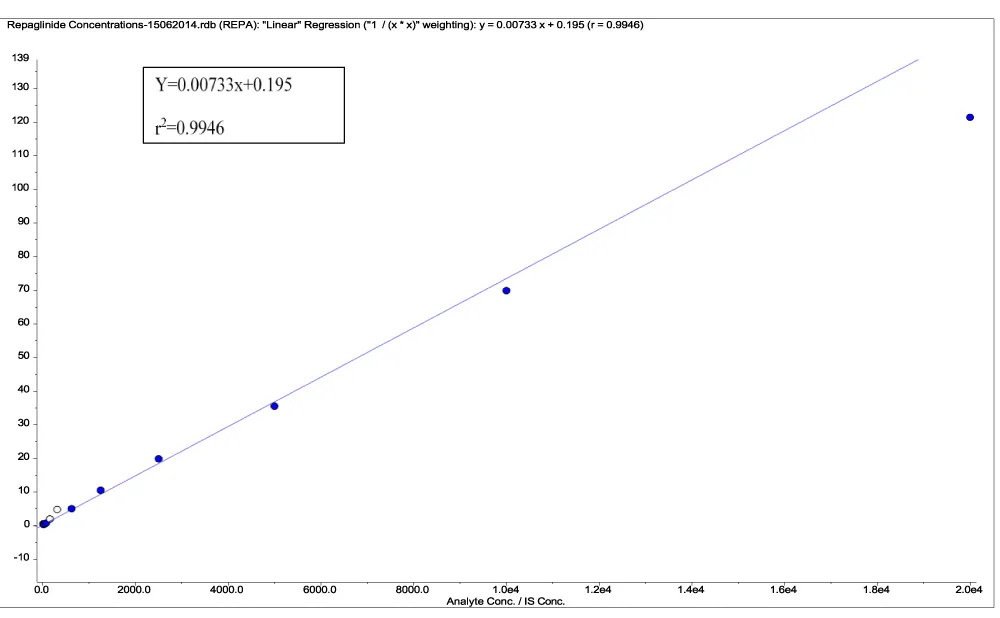
The analysis of plasma samples was conducted following a comprehensive validation of the LC-MS/MS methodology. No interfering peaks were detected in the chromatograms of blank plasma at the elution times corresponding to Indinavir and repaglinide, thereby demonstrating the selectivity of the employed method. The elution times for repaglinide and the internal standard were recorded at 3.56 and 3.47 minutes, respectively (Figure 1). Linearity was evaluated using nine calibration standards of repaglinide, a range of concentrations from 14.5 to 20,000 ng/mL. The correlation coefficient (r2) for repaglinide was determined to be 0.994 (Figure 2). Quality Control (QC) samples were prepared at low, medium, and high concentrations to assess accuracy, precision, and recovery. The intraday precision (within the same day) and interday precision (over three consecutive days), based on measurements of at least three replicates, were both less than 2% relative standard deviation, with a recovery rate of 98 to 102% in rat plasma (Table 1). The lower Limit Of Quantification (LOQ) and lower Limit Of Detection (LOD) were determined by injecting three replicates of 29 ng/mL based on the signal-to-noise ratio. The quantifiable LOQ was 9 ng/mL, and the detectable but not quantifiable LOD was 5 ng/mL.
Influence of indinavir on the pharmacokinetics and pharmacodynamics profiles of repaglinide
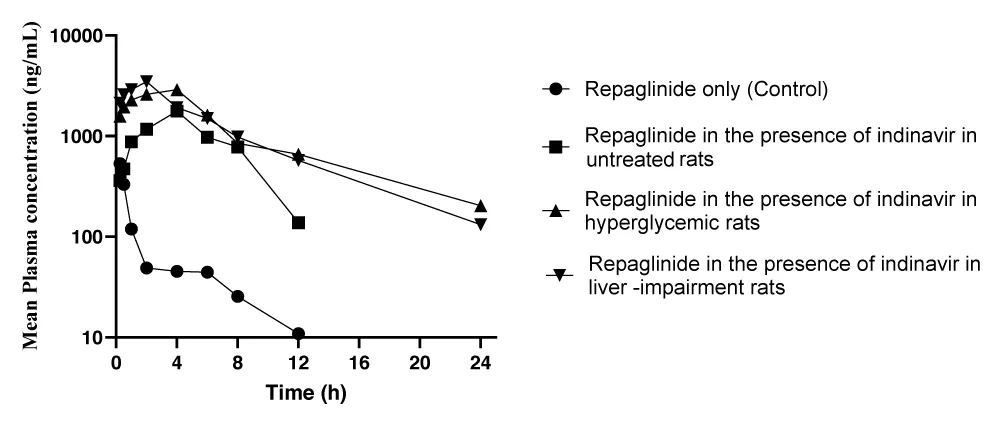
The mean plasma concentration of repaglinide was measured in healthy, diabetic, and hepatic-impaired rats, both with and without indinavir (Figure 3 and Table 2). Administration of a single dose of indanavir resulted in significant changes to the pharmacokinetic parameters of repaglinide, including maximum concentration (Cmax), Area Under the Curve (AUC), and total plasma clearance (CL), in all experimental groups relative to the group receiving repaglinide alone.TheCmax, AUC0-t, and CL found were 533 ng/mL, 678 ng/mL.h, and 11 ml/minute/kg respectively in group 1 healthy rats,1766 ng/mL, 10428 ng/mL.h, and 0.78 ml/minute/kg respectively in group 2 healthy rats, 2894 ng/mL, 23893 ng/mL.h, and 0.31 ml/minute/kg respectively in group 3 diabetic rats and 3481 ng/mL, 23395 ng/mL.h, and 0.31 ml/minute/kg respectively in group 4 hepatic impaired rats (Table 3). When compared to the repaglinide alone group, the Cmax of the remaining groups was increased by 3.3 to 6.5 fold. Similarly, AUC0-t was increased by 15.3 to 35.2 fold, and additionally, CL was reduced by 35.4 fold (Table 3).
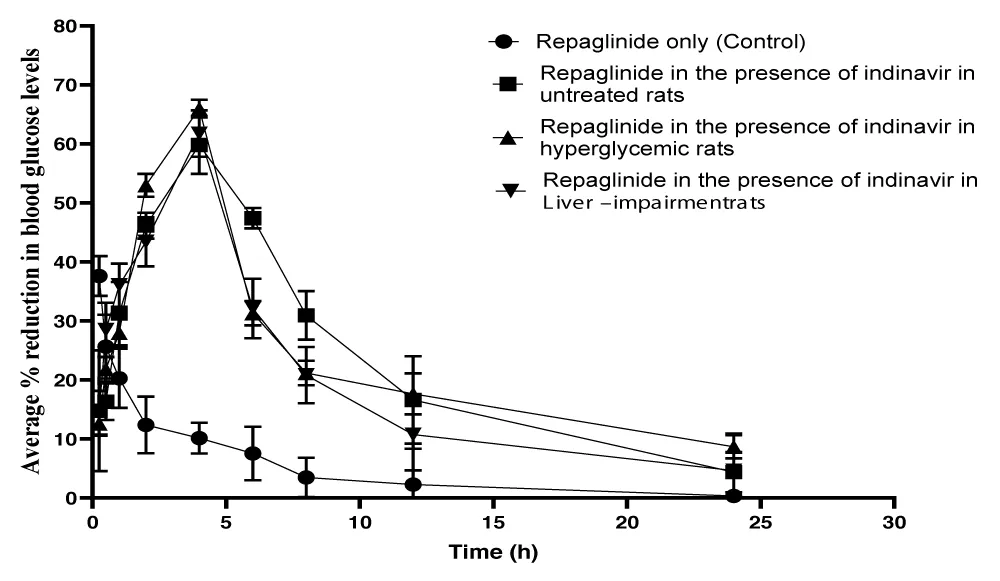
The hypoglycemic effect of repaglinide is attributed to its ability to stimulate the release of insulin from pancreatic beta cells. Serum samples were analyzed for blood glucose levels at various time intervals to calculate the percentage reduction in blood glucose levels. The results indicated a decreasing trend in glucose reduction for group 1, which ranged from 37.6% at 0.25 hours to 10.2% at 1 hour. In contrast, groups 2, 3, and 4 displayed an increasing trend in glucose reduction, with reductions ranging from 14.8% to 59.8% at intervals of 0.25 to 4 hours, 12.6% to 65.9% at 0.25 to 4 hours, and 14.4% to 61.7% at 0.25 to 4 hours, respectively (Figure 4 and Table 4).
Discussion
Patients infected with HIV often undergo polypharmacy, putting them at a heightened risk for drug-drug interactions. This creates prescribing challenges for clinicians managing HIV infections. Antiretroviral therapy has been linked to increased rates of insulin resistance, glucose intolerance, and diabetes mellitus, which presents a pharmacological challenge due to potential pharmacokinetic interactions between anti-diabetic and antiretroviral medications [18]. Most drug interactions in HIV treatment are pharmacokinetic, arising from changes in the Absorption, Distribution, Metabolism, and Excretion (ADME) profiles of either the HIV drug or the medications taken concurrently. These interactions may include modifications in drug metabolism mediated by the CYP-450 system, P-glycoprotein (P-gp), and Organic Anion Transporting Polypeptides (OATPs) modulation, which collectively regulate drug bioavailability and systemic exposure [19]. Antiretroviral drugs primarily undergo oxidative metabolism via the hepatic cytochrome P-450 system, particularly through the CYP3A4 isoform, which is the most prevalent in the human liver and plays a crucial role in metabolizing various medications [20].
Drug interactions with repaglinide have been noted, particularly with drugs that inhibit CYP 3A4, CYP2C8, and transporters such as OATP1B1 and P-gp, Repaglinide is primarily metabolized by CYP2C8, with a secondary contribution from CYP3A4, especially in intestinal first-pass metabolism, and is also a substrate for hepatic uptake transporter OATP1B1 [21].In addition to metabolic pathways, transporter proteins such as OATP1B1 play a crucial role in hepatic drug uptake. Pharmacovigilance data have indicated that certain antiretroviral agents, including indinavir, exhibit OATP1B1 inhibitory properties, which contributed to altered pharmacokinetics and increased systemic exposure of substrate drugs [22]. P-gp works alongside CYP3A4 and glutathione-S-transferase and may work synergistically to regulate the bioavailability of drugs administered orally. OATPs serve as membrane influx transporters that control the cellular uptake of various endogenous substances and important clinical drugs [23].
The plasma concentrations and percentage reduction in blood glucose levels of repaglinide were significantly altered in indinavir-treated rats, including normal, diabetic, and those with hepatic impairment, following a single dose administration. Indinavir notably increased the Cmax and AUC of repaglinide, indicating that it inhibited the CYP3A4-mediated metabolism of repaglinide, predominantly affecting first-pass metabolism in the intestine.
This suggests inhibition of both intestinal CYP3A4-mediated metabolism and transporter-mediated drug disposition (OATP1B1 and P-gp), leading to increased systemic exposure of repaglinide. Significant quantities of CYP3A4 are present in the mucosa of the small intestine, which plays a crucial role in drug interactions involving CYP3A4 inhibitors like indinavir.
The elevated plasma concentrations of repaglinide were noticeably greater in hepatic-impaired rats compared to normal and diabetic groups, indicating inhibited CYP 3A4 metabolism and altered activity of transporters (P-gp, OATP). This effect may be attributed to decreased hepatic metabolic capacity combined with impaired transporter function, resulting in reduced drug clearance and accumulation.
Compared to administration of repaglinide alone (control group), the half-life of repaglinide was markedly prolonged in untreated, hyperglycaemic, and liver-impaired rats co-treated with indinavir, accompanied by a reduction in repaglinide clearance. These findings indicate a decreased elimination of repaglinide, which is likely attributable to alterations in hepatic cytochrome P450 enzyme-mediated metabolism and OATP transporter activity. The enhanced plasma exposure of repaglinide was particularly pronounced in liver-compromised rats, potentially reflecting a combined effect of transporter-mediated suppression and compromised hepatic function. The observed enhancement in repaglinide bioavailability, as evidenced by increased Area Under the Curve (AUC) and maximum concentration (Cmax), is associated with elevated systemic drug levels and reduced elimination rates, as indicated by clearance and half-life parameters. Importantly, these pharmacokinetic alterations were associated with enhanced pharmacodynamic effects, as evidenced by a significant reduction in blood glucose levels. The increased systemic exposure of repaglinide was co-administered with indinavir likely prolonged insulin secretion from pancreatic β-cells through inhibition of ATP-sensitive potassium channels, thereby enhancing its glucose-lowering effect. However, this augmented pharmacological response may also increase the risk of hypoglycemia due to drug accumulation and delayed elimination.
From a clinical perspective, these findings are highly relevant in HIV-infected patients receiving indinavir-based antiretroviral therapy. Co-administration of repaglinide in such patients may necessitate dose adjustment and careful therapeutic monitoring to avoid severe hypoglycemic events [24].
Kinetic investigations employing human liver microsomes have demonstrated that indinavir functions as a reversible suppressor of cytochrome P-450 enzymes. Notably, no time-dependent diminution of enzymatic activity was detected upon incubation of human liver microsomes with indinavir and NADPH. Additional examination of indinavir’s impact on testosterone 6β-hydroxylation by human liver microsomes indicated that indinavir acts as a competitive antagonist, with an inhibition constant (Ki) of 0.5 mM [25]. Noted that all clinically available HIV protease suppressors can interact with numerous drugs through competition for CYP3A4, and indinavir could inhibit drugs metabolized by this enzyme [26,27]. Highlighted that co-administering repaglinide with a well-established P-glycoprotein suppressor cyclosporine significantly elevated the plasma concentration levels of repaglinide; this effect is likely due to inhibition of repaglinide metabolism by CYP3A4 and decreased hepatic uptake mediated through OATP1B1. Consequently, cyclosporine might amplify the antidiabetic action of repaglinide and elevate the risk of hypoglycemia in humans [27]. Our findings align with those by [28], which demonstrated that clarithromycin, a known CYP3A4 blocker, markedly increased the AUC0-∞ and Cmax of repaglinide, enhancing its blood glucose-lowering impact [29]. Furthermore, [27] noted that both gemfibrozil and atorvastatin led to a significant rise in the AUC0-∞ of repaglinide, consequently enhancing its hypoglycemic effects [30].
Conclusion
This study found that indinavir increased the bioavailability of repaglinide by inhibiting CYP3A4-facilitated metabolism (both intestinal and hepatic). Additionally, it hinders the hepatic OATP transport activity, mediated by Indinavir. The observed effect was evident across absorption (first pass effect), metabolic (liver-mediated), and excretion (OATP transporter suppression) stages. This research highlights the risk of potential drug interactions when repaglinide is used alongside indinavir in populations such as those who are normal, diabetic, or have hepatic impairments. Consequently, the concomitant use of repaglinide and indinavir with caution or avoided in clinical practice. Given the clinical implications, further investigation through controlled clinical trials is warranted to elucidate the extent and impact of this interaction.
Acknowledgments
The authors acknowledge the support and research facilities provided by Acubiosys Private Limited, Hyderabad, for the successful completion of this study.
Research ethics
All animal experiments were conducted in accordance with institutional ethical guidelines and approved by the IAEC approval number (1305/ac/09/CPCSEA).
Author contributions
M. Jeevan Karthik and J. Lokesh designed the study, performed dosing, and analyzed the data. T. Aakarsha and Janga Ramesh Babu prepared doses and contributed to manuscript drafting. C. Manaswini assisted with references and manuscript drafting. Dr. Srinivas Maddi supervised the study and reviewed the final manuscript. All authors read and approved the final manuscript.
Conflict of interest: The authors declare no conflict of interest.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
- Cho CK, Ko E, Mo JY, Kang P, Jang CG, Lee SY, et al. PBPK modeling to predict the pharmacokinetics of pantoprazole in different CYP2C19 genotypes. Arch Pharm Res. 2024;47(1):82-94. Available from: https://doi.org/10.1007/s12272-023-01478-7
- Nasution MAH, Nasution MS. Correlation between duration of protease inhibitor therapy with insulin resistance in HIV patients. J Endocrinol Trop Med Infect Dis. 2020;2(2):96-106. Available from: https://doi.org/10.32734/jetromi.v2i2.3875
- Mastan SK, Chaitanya G, Raghunandan Reddy K, Eswar Kumar K. Possible metabolic interactions between antiretroviral drugs and antidiabetic drugs: an overview. Int J PharmTech Res. 2009;1(3):613-622. Available from: https://www.researchgate.net/publication/255607354_Possible_metabolic_interactions_between_antiretroviral_drugs_and_antidiabetic_drugs_An_overview
- Mettu VS, Swami PY, Abigna P, Nath AR, Sharma G. In vitro and in vivo (mouse) evaluation of drug-drug interactions of repaglinide with anti-HIV drugs. Pharmacol Pharm. 2015;6(4):241-246. Available from: http://dx.doi.org/10.4236/pp.2015.64026
- Khedkar JP, Salunke PS, Mahajan HD, Shaikh TJ, Bagul MA, Patil UH. Effects of amiodarone on the glucose-lowering effect of glimepiride: an animal experiment. Uttar Pradesh J Zool. 2024;45(19):189-195. Available from: https://doi.org/10.56557/upjoz/2024/v45i194516
- Li Y, Li X, Zhu M, Liu H, Lei Z, Yao X, et al. Development of a physiologically based pharmacokinetic population model for diabetic patients and its application to understand disease-drug–drug interactions. Clin Pharmacokinet. 2024;63(6):831-845. Available from: https://doi.org/10.1007/s40262-024-01383-2
- Suresh DK, Vibhute A, Uddin IMD. Screening the influence of diltiazem on oral antidiabetic agents in experimental animals. Indian Drugs. 2022;59(3):54-61. Available from: https://www.researchgate.net/publication/361167674_SCREENING_THE_INFLUENCE_OF_DILTIAZEM_ON_ORAL_ANTIDIABETIC_AGENTS_IN_EXPERIMENTAL_ANIMALS
- Atta MG, De Seigneux S, Lucas GM. Clinical pharmacology in HIV therapy. Clin J Am Soc Nephrol. 2019;14(3):435-444. Available from: https://doi.org/10.2215/cjn.02240218
- Choi DH, Choi JS. Effects of amlodipine on the pharmacokinetics of repaglinide. Korean J Clin Pharm. 2011;21.
- Mastan SK, Eswar Kumar K. Influence of indinavir on the pharmacodynamic activity of gliclazide in rats and rabbits. Int J Integr Biol. 2009:16-21. Available from: https://www.researchgate.net/publication/44259440_Influence_of_indinavir_on_the_pharmacodynamic_activity_of_gliclazide_in_rats_and_rabbits
- Bhattachar SN, Bender DM, Sweetana SA, Wesley JA. Discovery formulations: approaches and practices in early preclinical development. 2015. Available from: https://link.springer.com/chapter/10.1007/978-1-4939-1399-2_2
- Jo EJ, Bae E, Yoon JH, Kim JY, Han JS. Comparison of murine retroorbital plexus and facial vein blood collection to mitigate animal ethics issues. Lab Anim Res. 2021;37(1). Available from: https://doi.org/10.1186/s42826-021-00090-4
- Nagaraju B, Anilkumar KV. Pharmacodynamic and pharmacokinetic interaction of losartan with glimepiride-metformin combination in rats and rabbits. Indian J Pharmacol. 2021;53(6):465-470. Available from: https://doi.org/10.4103/ijp.ijp_845_19
- Raafat K, Aboul-Ela M, El-Lakany A. Alloxan-induced diabetic thermal hyperalgesia, prophylaxis, and phytotherapeutic effects of Rheum ribes L. in mouse model. Arch Pharm Res. 2021;44(8):1-10. Available from: https://doi.org/10.1007/s12272-014-0372-y
- Robin S, Sunil K, Nidhi S. Different models of hepatotoxicity and related liver diseases: a review. Int Res J Pharm. 2012;3(7):86-95. Available from: http://www.irjponline.com
- Shaik B, Eerla A, Palatheeya S, Veerareddy PR, Maddi S. A validated HPLC method for the simultaneous quantification of curcumin, silybin, and psoralen: application to phytopharmaceuticals. Rev Bras Farmacogn. 2023;33(3):610-616. Available from: https://link.springer.com/article/10.1007/s43450-023-00392-9
- Neerati P, Gade J. Influence of atorvastatin on the pharmacokinetics and pharmacodynamics of glyburide in normal and diabetic rats. Eur J Pharm Sci. 2011;42(3):285-289. Available from: https://doi.org/10.1016/j.ejps.2010.12.006
- Lin JH, Chiba M, Chen IW, Nishime JA, deLuna FA, Yamazaki M, et al. Effect of dexamethasone on the intestinal first-pass metabolism of indinavir in rats: evidence of cytochrome P-450 and P-glycoprotein induction. Drug Metab Dispos. 1999;27(10):1187-1193. Available from: https://pubmed.ncbi.nlm.nih.gov/10497146/
- Loos NHC, Beijnen JH, Schinkel AH. The mechanism-based inactivation of CYP3A4 by ritonavir: what mechanism? Int J Mol Sci. 2022;23(17). Available from: https://doi.org/10.3390/ijms23179866
- Hirlekar RS, Momin AM. Advances in drug delivery from nose to brain: an overview. Curr Drug Ther. 2018;13(1):4-24. Available from: http://dx.doi.org/10.2174/1574885512666170921145204
- Hakkola J, Hukkanen J, Turpeinen M, Pelkonen O. Inhibition and induction of CYP enzymes in humans: an update. Arch Toxicol. 2020;94(11):3671-3722. Available from: https://doi.org/10.1007/s00204-020-02936-7
- Stäuble CK, Toma V, Stammschulte T, Allemann SS, Meyer zu Schwabedissen HE. Prevalence of substances with OATP1B1 inhibitory properties in individual case safety reports of suspected statin-associated myopathy: an analysis of Swiss pharmacovigilance data. Eur J Clin Pharmacol. 2026;82(3). Available from: https://link.springer.com/article/10.1007/s00228-025-03968-7
- Sharma K, Pawar G, Yadam S, Giri S, Rajagopal S, Mullangi R. LC-MS/MS-ESI method for simultaneous quantitation of metformin and repaglinide in rat plasma and its application to pharmacokinetic study in rats. Biomed Chromatogr. 2013;27(3):356-364. Available from: https://doi.org/10.1002/bmc.2799
- Chiba M, Hensleigh M, Lin JH. Hepatic and intestinal metabolism of indinavir, an HIV protease inhibitor, in rat and human microsomes: major role of CYP3A. Biochem Pharmacol. 1997;53(8):1187-1195. Available from: https://doi.org/10.1016/s0006-2952(97)00100-7
- Zhou S, Zeng S, Shu Y. Drug-drug interactions at organic cation transporter 1. Front Pharmacol. 2021;12. Available from: https://doi.org/10.3389/fphar.2021.628705
- Ji C. Molecular factors and pathways of hepatotoxicity associated with HIV/SARS-CoV-2 protease inhibitors. Int J Mol Sci. 2023;24(9). Available from: https://doi.org/10.3390/ijms24097938
- Gelotte CK, Albrecht HH, Hynson J, Gallagher V. A multicenter, randomized, placebo-controlled study of pseudoephedrine for the temporary relief of nasal congestion in children with the common cold. J Clin Pharmacol. 2019;59(12):1573-1583. Available from: https://doi.org/10.1002/jcph.1472
- Niemi M, Neuvonen PJ, Kivistö KT. The cytochrome P4503A4 inhibitor clarithromycin increases the plasma concentrations and effects of repaglinide. Clin Pharmacol Ther. 2001;70(1):58-65. Available from: https://doi.org/10.1067/mcp.2001.116511
- Pakkir Maideen NM, Manavalan G, Balasubramanian K. Drug interactions of meglitinide antidiabetics involving CYP enzymes and OATP1B1 transporter. Ther Adv Endocrinol Metab. 2018;9(8):259-268. Available from: https://doi.org/10.1177/2042018818767220
- Rodríguez-Antona C, Jande M, Rane A, Ingelman-Sundberg M. Identification and phenotype characterization of two CYP3A haplotypes causing different enzymatic capacity in fetal livers. Clin Pharmacol Ther. 2005;77(4):259-270. Available from: https://doi.org/10.1016/j.clpt.2004.11.003
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
